emDOCs.net – Emergency Medicine EducationTOXCard: Loperamide Toxicity - emDOCs.net - Emergency Medicine Education

Daniel Buchen on X: "Challenging myself to make one #FOAMed post from now until the end of Sept. This first one is a mnemonic I invented to help myself remember the most

The 2023 Canadian Cardiovascular Society Clinical Practice Update on Management of the Patient With a Prolonged QT Interval - Canadian Journal of Cardiology
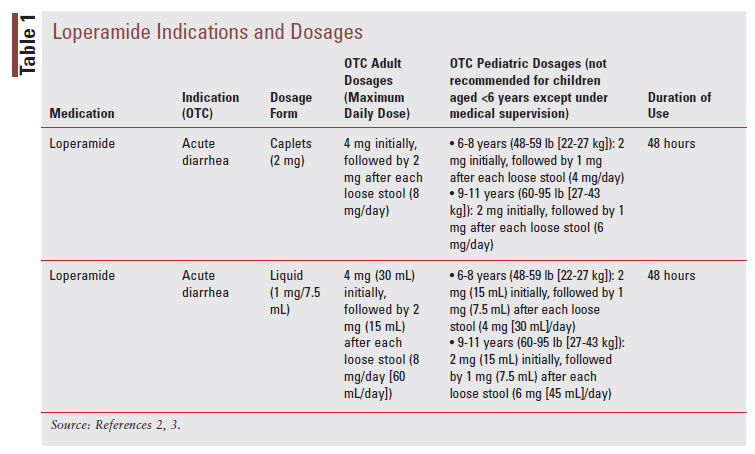
![PDF] Identification of Patients At-Risk of QT Interval Prolongation during Medication Reviews: A Missed Opportunity? | Semantic Scholar PDF] Identification of Patients At-Risk of QT Interval Prolongation during Medication Reviews: A Missed Opportunity? | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/0f81d7ab96eb51e3f7101fa71db7f95142bcac25/3-Table1-1.png)
PDF] Identification of Patients At-Risk of QT Interval Prolongation during Medication Reviews: A Missed Opportunity? | Semantic Scholar

Prevalence of QTc Prolongation in Patients With Advanced Cancer Receiving Palliative Care—A Cause for Concern? - ScienceDirect

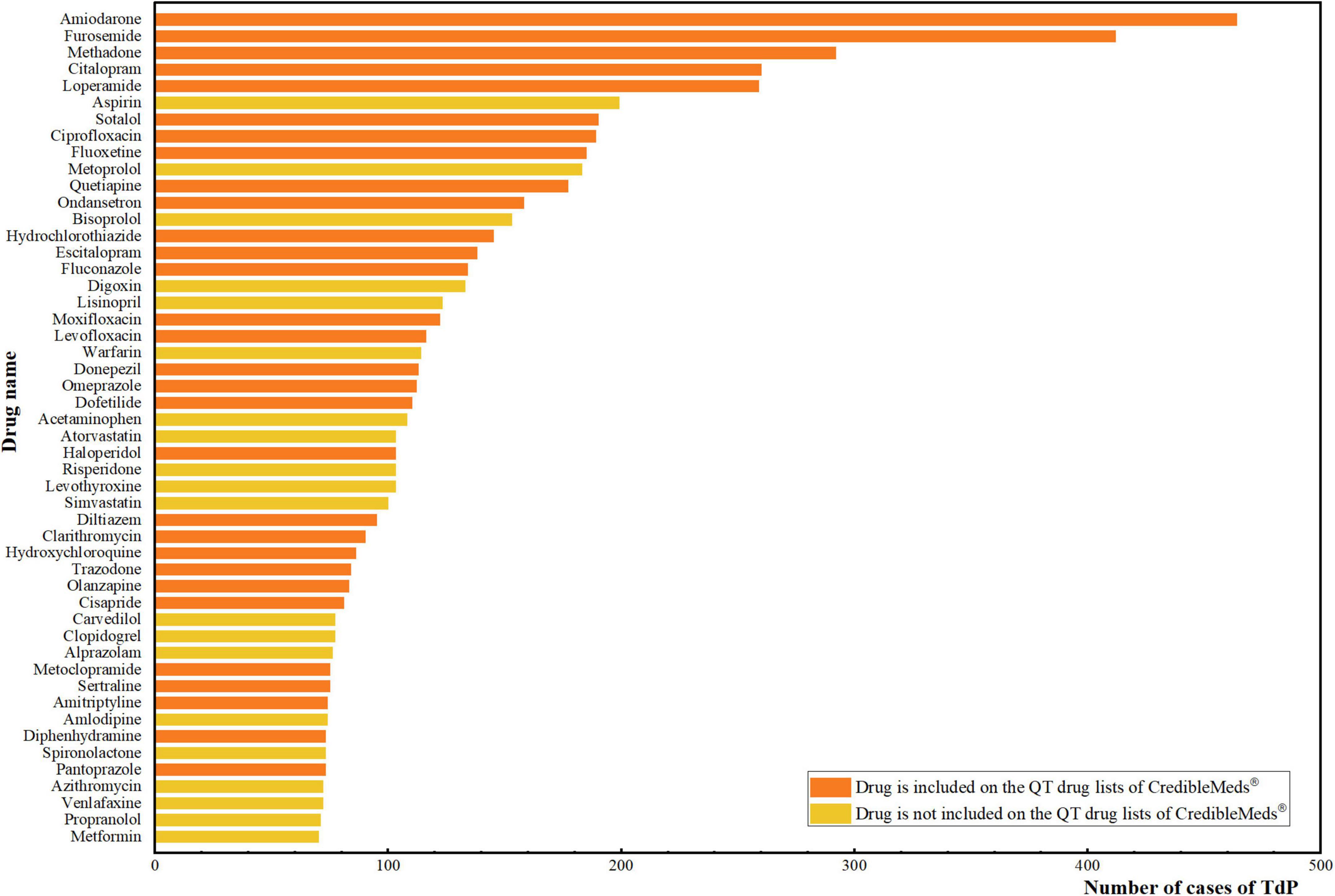
Proportional Reporting Ratios for QTc-Prolongation/Torsade de Pointes,... | Download Scientific Diagram
33469-0.fp.png)
THE LONG-LASTING HIGH: CARDIAC COMPLICATIONS FROM LOPERAMIDE TOXICITY | Journal of the American College of Cardiology

A Case Report of Loperamide-Induced Ventricular Storm - Jerome De Vera, Hyungjin Ben Kim, Antoine E. Sakr, 2021
33247-4.fp.png)
LOPERAMIDE CARDIOTOXICITY: A BYPRODUCT OF THE OPIOID EPIDEMIC | Journal of the American College of Cardiology

A Case Report of Loperamide-Induced Ventricular Storm - Jerome De Vera, Hyungjin Ben Kim, Antoine E. Sakr, 2021

Mechanisms and management of drug‐induced QT prolongation - Parsons - 2022 - Prescriber - Wiley Online Library
Wide interindividual variability in cardiovascular toxicity of loperamide: A case report and review of literature

Sinus rhythm and prolonged QTc of 472 msec, several days after stopping... | Download Scientific Diagram

Loperamide cases reported to United States poison centers, 2010–2022 | Injury Epidemiology | Full Text